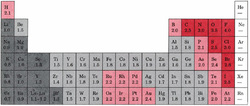
Electronegativity can be understood as chemical
property describing an atom's ability to attract and bind to electrons. Because
electronegativity is a qualitative property, there is not a standardized method
for calculating electronegativity. However, the scale that most chemists use in
quantifying electronegativity is the Pauling Scale, named after the chemist
Linus Pauling. The numbers assigned by the Pauling scale are dimensionless due
to electronegativity being largely qualitative. Electronegativity values for
each element can be found on certain periodic tables. An example is provided
below.
property describing an atom's ability to attract and bind to electrons. Because
electronegativity is a qualitative property, there is not a standardized method
for calculating electronegativity. However, the scale that most chemists use in
quantifying electronegativity is the Pauling Scale, named after the chemist
Linus Pauling. The numbers assigned by the Pauling scale are dimensionless due
to electronegativity being largely qualitative. Electronegativity values for
each element can be found on certain periodic tables. An example is provided
below.
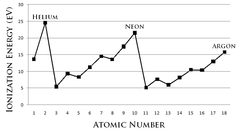
Ionization Energy is the
amount of energy required to remove an electron from a neutral atom in its
gaseous phase. Conceptually, ionization energy is considered the opposite of
electronegativity. The lower this energy is, the more readily the atom becomes a
cation. Therefore, the higher this energy is, the more unlikely the atom becomes
a cation. Generally, elements on the right side of the periodic table have a
higher ionization energy because their valence shell is nearly filled. Elements
on the left side of the periodic table have low ionization energies because of
their willingness to lose electrons and become cations. Thus, ionization energy
increases from left to right on the periodic table.
amount of energy required to remove an electron from a neutral atom in its
gaseous phase. Conceptually, ionization energy is considered the opposite of
electronegativity. The lower this energy is, the more readily the atom becomes a
cation. Therefore, the higher this energy is, the more unlikely the atom becomes
a cation. Generally, elements on the right side of the periodic table have a
higher ionization energy because their valence shell is nearly filled. Elements
on the left side of the periodic table have low ionization energies because of
their willingness to lose electrons and become cations. Thus, ionization energy
increases from left to right on the periodic table.
Electron Affinity
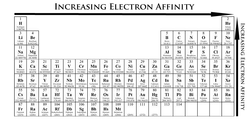
electron affinity describes the ability of an atom to accept an electron. Unlike
electronegativity, electron affinity is a quantitative measure that measures the
energy change that occurs when an electron is added to a neutral gas atom. When
measuring electron affinity, the more negative the value, the more of an
affinity to electrons that atom has.
electronegativity, electron affinity is a quantitative measure that measures the
energy change that occurs when an electron is added to a neutral gas atom. When
measuring electron affinity, the more negative the value, the more of an
affinity to electrons that atom has.
Atomic Radius

the atomic radius is one-half the distance between the nuclei of two atoms is
(just like a radius is half the diameter of a circle). However, this idea is
complicated by the fact that not all atoms are normally bound together in the
same way. Some are bound by covalent bonds in molecules, some are attracted to
each other in ionic crystals, and others are held in metallic crystals.
Nevertheless, it is possible for a vast majority of elements to form covalent
molecules in which two like atoms are held together by a single covalent bond.
The covalent radius of these molecules is often referred to as the atomic
radius. This distance is measured in picometers. Going through each of the
elements of the periodic table, patterns of the atomic radius can be seen.
(just like a radius is half the diameter of a circle). However, this idea is
complicated by the fact that not all atoms are normally bound together in the
same way. Some are bound by covalent bonds in molecules, some are attracted to
each other in ionic crystals, and others are held in metallic crystals.
Nevertheless, it is possible for a vast majority of elements to form covalent
molecules in which two like atoms are held together by a single covalent bond.
The covalent radius of these molecules is often referred to as the atomic
radius. This distance is measured in picometers. Going through each of the
elements of the periodic table, patterns of the atomic radius can be seen.
Metalic

The metallic character of an element can be defined as how readily an atom can
lose an electron. As you move from right to left across a period, metallic
character increases because
the attraction between valence electron and the nucleus is weaker,
thus enabling an easier loss of electrons. Metallic character increases as you
move down a group because the atomic
size is increasing. When the atomic size increases, the outer shells are farther
away. The principle
quantum number increases and average electron density moves farther from
nucleus. The
electrons of the valence shell have less of an attraction to the nucleus and, as
a result, can lose electrons more readily, causing an increase in metallic
character.
lose an electron. As you move from right to left across a period, metallic
character increases because
the attraction between valence electron and the nucleus is weaker,
thus enabling an easier loss of electrons. Metallic character increases as you
move down a group because the atomic
size is increasing. When the atomic size increases, the outer shells are farther
away. The principle
quantum number increases and average electron density moves farther from
nucleus. The
electrons of the valence shell have less of an attraction to the nucleus and, as
a result, can lose electrons more readily, causing an increase in metallic
character.
Notes
Atomic radius increases going down a colum. Atomic radius decreases left to right.
Anion will be larger than there parent atom.Cation will be smaller
Decreases as you go down the table. increases as it goes to the right.
Covalent bond-share equally
polar covalent bond- unequally shared
ioinic bond- not shared
Ionization decreases down the colums. increases across a period.
gets harder to take an electron the higher the ionization energy is.
Anion will be larger than there parent atom.Cation will be smaller
Decreases as you go down the table. increases as it goes to the right.
Covalent bond-share equally
polar covalent bond- unequally shared
ioinic bond- not shared
Ionization decreases down the colums. increases across a period.
gets harder to take an electron the higher the ionization energy is.
Atomic theory
John Dalton: "Experimental Essays" on the constitution of mixed gases; on the pressure of steam and other vapours
at different temperatures, both in a vacuum and in air;
on evaporation; and on the thermal expansion of gases After describing experiments to ascertain the pressure of steam at various
points between 0 and 100 °C (32 and 212 °F), Dalton concluded from observations
on the vapour pressure of six different liquids, that
the variation of vapour pressure for all liquids is equivalent, for the same
variation of temperature, reckoning from vapour of any given pressure
JJ Thompson:He is credited with discovering electrons and isotopes
Ernest rutherford: he discovered the concept of radioactive half-life, proved that radioactivity involved the
transmutation of one chemical element to another, and also differentiated and
named alpha and beta radiation,[3]
proving that the former was essentially helium ions
Ernest Rutherford:he theorized that atoms have their charge concentrated in a very small nucleus,[6] and
thereby pioneered the Rutherford model of the atom, through his discovery and interpretation of
Rutherford scattering in his gold foil experiment.
Niels Bohr: He developed the model of the atom with the nucleus at the center and electrons
in orbit around it, which he compared to the planets orbiting the sun.[3] He
worked on the idea in quantum mechanics that electrons move from one energy
level to another in discrete steps, not continuously.
at different temperatures, both in a vacuum and in air;
on evaporation; and on the thermal expansion of gases After describing experiments to ascertain the pressure of steam at various
points between 0 and 100 °C (32 and 212 °F), Dalton concluded from observations
on the vapour pressure of six different liquids, that
the variation of vapour pressure for all liquids is equivalent, for the same
variation of temperature, reckoning from vapour of any given pressure
JJ Thompson:He is credited with discovering electrons and isotopes
Ernest rutherford: he discovered the concept of radioactive half-life, proved that radioactivity involved the
transmutation of one chemical element to another, and also differentiated and
named alpha and beta radiation,[3]
proving that the former was essentially helium ions
Ernest Rutherford:he theorized that atoms have their charge concentrated in a very small nucleus,[6] and
thereby pioneered the Rutherford model of the atom, through his discovery and interpretation of
Rutherford scattering in his gold foil experiment.
Niels Bohr: He developed the model of the atom with the nucleus at the center and electrons
in orbit around it, which he compared to the planets orbiting the sun.[3] He
worked on the idea in quantum mechanics that electrons move from one energy
level to another in discrete steps, not continuously.
